INTRODUCTION: This study was performed to determine the survival rate of various organisms in the test product. The test employed methods designed to determine antimicrobial effectiveness described in the United States Pharmacopeia (USP).
The samples of the product were inoculated, with five standard test organisms. The inoculated samples were then incubated for a total of 28 days at 20-25°C. Aliquots from the samples were immediately removed and assayed for surviving organisms at 0 hour, 7 day, 14 day, and 28 day time intervals. The log reduction in the level of the test organisms was calculated for each time interval.
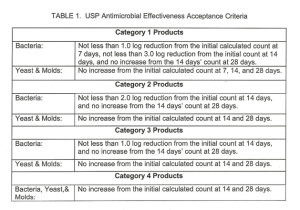
ACCEPTANCE CRITERIA: According to USP, products are placed into categories according to product type. The product category will determine the acceptance criteria for the test. The following table provides a description of the current USP categories.
CATEGORY PRODUCT DESCRIPTION Injections, other parenterals including emulsions, otic products,
- sterile nasal products, and ophthalmic products made with aqueous bases or vehicles. Topically used products made with aqueous bases or vehicles, non
- sterile nasal products, and emulsions, including those applied to mucous membranes.
- oral products other than antacids, made with aqueous bases or vehicles.
- antacids made with an aqueous base.
The acceptance criteria for the USP Antimicrobial Effectiveness Test is located in Table 1. No increase is defined as not more than a 0.5 10glOunit higher than the previous value measured.
PROCEDURE:
The following organisms were tested:
- Staphylococcus aureus ATCC #6538 [Bacteria, Gram (+) cocci]
- Pseudomonas aeruginosa ATCC #9027 [Bacteria, Gram (-) bacillus]
- Escherichia coli ATCC #8739 [Bacteria, Gram (-) bacillus]
- Candida albicans ATCC #10231 [Yeast]
- Aspergillus niger ATCC #16404 [Mold]
The bacteria were transferred to soybean casein digest agar (SCDA) and incubated at fO-35°C for 18-24 hours. The C. albicans and A. niger were transferred to Sabouraud dextr9se agar (SDEX). The C. albicans was incubated at 20-25°C for 44-52 hours and the A. niger was incubated at 20-25°C for 6-10 days.
The bacteria and C. albicans were harvested using 0.9% saline (PHSS) and A. niger was harvested with 0.9% saline containing 0.05% polysorbate 80 (Tween® 80). The A. niger was filtered through sterile gauze and all cultures were vortexed to break up clumps. After harvesting, the organisms were centrifuged and re-suspended in purified water (PURW). Each suspension was adjusted to approximately 108 colony forming units (CFU)/mL using visual turbidity. The actual concentration of each culture was determined using the positive control values.
These results represent duplicate analysis plated in triplicate. Individual samples of the test product were prepared for each challenge organism. The tubes containing the sample were inoculated with the test organisms using a calibrated micropipettor. The volume of the inoculum was between 0.5% and 1.0% of the volume of the product. The final concentration of the test preparation was approximately 105 – 10 6 CFU/mL of product. The samples were well mixed.
Positive control tubes were prepared for each organism using sterile water. The volume used for the positive control was equivalent to that used in the samples. Negative controls were also prepared. The positive controls were then inoculated in the same manner as the test samples. All test samples were stored at 20-25°C for a total of 28 days.
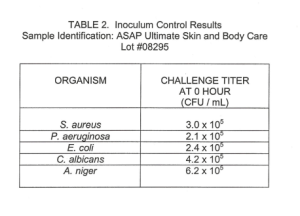
Control tubes and test vials were assayed immediately to determine the initial concentration of organisms in each tube. The test suspensions were assayed at the following intervals: 0 hour, 7, 14, and 28 days.
Sample aliquots at each interval were diluted in Letheen broth (LETH) and plated on SCDA for bacteria and SDEX for C. albicans and A. niger. The bacteria plates were incubated at 30-35°C for 3-5 days. The C. albicans plates were incubated at 20-25°C for 3-5 days and the A. niger plates were incubated at 20-25°C for 3-7 days.
RESULTS:
The results are reported in Tables 2-3. The greater than (» values represent the detectable limits of the test where zero CFU were observed on the plates. The approximate (-) symbol is applied to results where plate counts fell outside of the statistically accurate range of 25-250 CFU for bacteria and yeast and 8-80 CFU for mold.
Qualification of neutralization was not performed. Sponsor is responsible for proving neutralization of the product.
All negative controls showed no growth. Testing met the acceptance criteria stated in this report.
CONCLUSION:
Interpretation of the data is the responsibility of the sponsor and no conclusion can be made by Nelson Laboratories, Inc. (NLI).
DATA DISPOSITION:
The raw data and final report from this study are archived at NLI or an approved off-site location.
STATEMENT OF UNCERTAINTY:
If applicable, the statement of uncertainty is available to sponsors upon request.

All reports and letters issued by Nelson Laboratories, Inc. are for the exclusive use of the sponsor to whom they are addressed. These results relate only to the samples tested. Reports may not be reproduced except in their entirety. No quotations from reports or use of the corporate name is permitted except as expressly authorized by Nelson Laboratories, Inc. in writing. The significance of any data is subject to the adequacy and representative character of the samples tendered for testing. Nelson Laboratories, Inc. warrants that all tests are performed in accordance with established laboratory procedures and standards. Nelson Laboratories, Inc. makes no other warranties of any kind, express or implied. Nelson Laboratories, Inc. expressly states that it makes no representation or warranty regarding the adequacy of the samples tendered for testing for any specific use of application, that determination being the sole responsibility of the sponsor. Nelson Laboratories’ liability for any loss or damage resulting from its actions or failure to act shall not exceed the cost of tests performed, and it shall not be liable for any incidental or consequential damages.


